Validating the Effectiveness of an Optical Surgical Navigator

The validation of the effectiveness of an optical surgical navigator is a topic that has a vital importance for the surgical practice. Currently, optical surgical browsers are a consolidated technology and have become a crucial tool to improve accuracy and safety in a large number of surgical procedures….
What are the steps to use and calibrate a meter?

It is important to follow the instructions and recommendations of manufacturers for the use and calibration of a phmeter, since, to achieve a correct measurement with a phmeter must be calibrated correctly, remember that the pH measures the concentration of hydrogen ions, which determine the degree of acidity and basicity of a solution; in addition not only works to measure the degree of water contamination, but as a diagnosis of body diseases or when measuring pH of urine and blood.
Use of the PH meter to determine the degree of reduction of a sample

The pH meter is used to determine the degree of decline of a sample. This is important because it indicates the amount of acid or base in the sample and determines the degree of contamination. Likewise, it measures the concentration of H+ ions in a sample, where it is directly related to the alkalinity of the sample, the higher the concentration of ions, the more acidic the sample. In contrast, the lower the ion concentration, the more basic the sample.
pH meter, water pH, milk pH, and human body pH
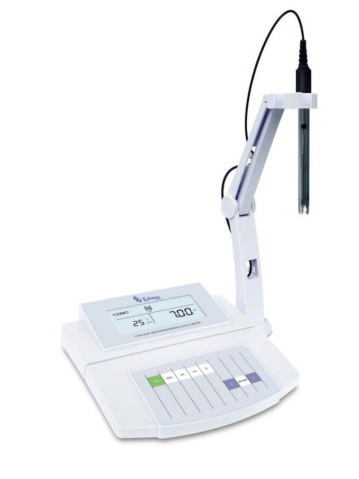
PH is a parameter used to measure the concentration of the hydrogen ion [H +] in a solution. In definition pH, it is the negative logarithm of base 10 of the activity of the [H +]. PH values vary on a scale from 0 to 14, which indicates the degree of acidity or alkalinity of an aqueous solution.
pH meter or pH meter: How should it be used?

A pH meter is an instrument used to measure the acidity or alkalinity of a solution. The information provided by this equipment expresses the degree of acidity of an acid or base in terms of the activity of hydrogen ions.
What is pH and how is it measured?
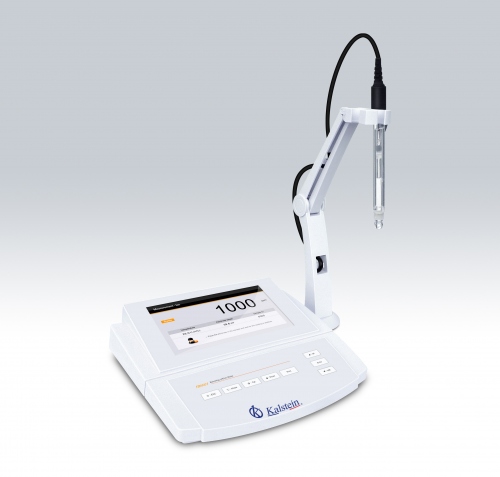
pH is the unit of measurement that describes the degree of acidity or alkalinity and is measured on a scale that goes from 0 to 14. The measurement of pH (hydrogen potential) is one of the most important parameters in a laboratory, it has a great field of application such as: research, industry, teaching, among others.
How does a pH meter work?

A pH meter or pH meter is an instrument used to measure the acidity or alkalinity of a solution. The information provided by this equipment expresses the degree of acidity of an acid or a base in terms of the activity of the hydrogen ions.
Importance of pHmeter in a laboratory

A pH meter is one of the most used equipment in a laboratory. It is used to measure the acidity or alkalinity of a solution and the information it provides expresses the degree of acidity of an acid or base in terms of the activity of hydrogen ions, so through the use of this equipment we can know the type of substance we are analyzing, as well as the type of treatment that should be given to it.
Use of the PH meter for the determination of the oxidation degree of a sample
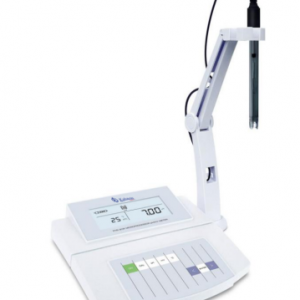
The pH meter is a very useful tool to determine the degree of oxidation of a sample. In most cases, it is used to measure the degree in liquids and solids. The sample can be measured with a portable device, according to a benchtop pH meter. The accuracy of the measurement depends on the type of table or pocket.
Surgical browsers: What technology do they use?

A surgical navigator is a sophisticated medical team that is employed in the precise planning and execution of surgical procedures in operating rooms, that is, they act as a guide to other surgical instruments such as the scalpel. Its structure consists of a series of instruments that are connected to screens by sensors
